
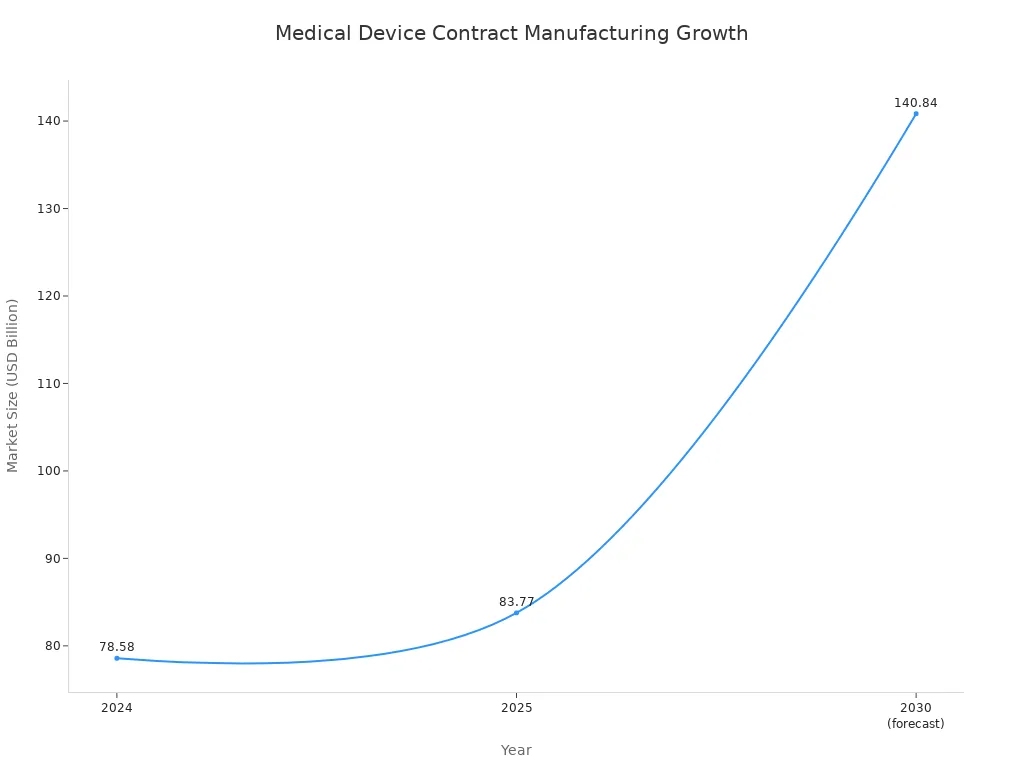
The medical device manufacturing industry is experiencing rapid growth, primarily due to the continuous emergence of new technologies and the growing market demand for safe products. Given that consumers are always looking for reliable products, the global medical device contract manufacturing market reached US$76.8 billion in 2024. Experts predict that this strong growth momentum will continue until 2030.
- Regulations help keep patients safe and bring new ideas.
- Agencies like the FDA support safety and new inventions.
- Flexible rules help people use new tech, like AI, in devices.
Picking the right technologies and materials makes safe manufacturing easier than ever. It also makes the process simple.
Technologies in Medical Device Manufacturing
A new wave of cutting-edge tools is revolutionizing how medical devices are made. This isn’t just an upgrade—it’s a game-changer. Companies are now empowered to build safer and fully personalized products, fundamentally shifting how the entire industry operates.

Automation and Robotics
Automation and robotics are changing the way many manufacturing devices operate. Robotic arms can perform tasks that require precision and careful observation. They often work in clean rooms to ensure that products remain clean. Sometimes, collaborative robots also work alongside humans to package and wrap items. This collaboration not only reduces errors but also significantly improves work efficiency.
Here is how robots help:
| Technology | Efficiency and Error Reduction Benefits |
| Cleanroom Robotics | Work in clean spaces, lowering germs and mistakes. |
| SCARA & Six-Axis Robots | Build and pack devices quickly and correctly, making less waste. |
| Machine Vision Systems | Check parts right away, finding problems before patients get them. |
| Flex Feeding Systems | Line up small parts for robots, lowering human mistakes. |
| Integration & Support | Custom help and training keep things running well and safe. |
Artificial intelligence-driven analytical tools also play an important role in the medical device manufacturing process. They can predict when equipment needs maintenance and assist engineers in planning their work. This ensures that factories run smoothly and saves a lot of money. In addition, automation technology helps companies comply with regulations and ensures that high-quality products are produced every time.

3D Printing
3D printing opens up new possibilities for medical device manufacturing. NOBLE engineers can create complex shapes that were previously difficult to achieve. Companies often use this technology to produce specialized implants, tools, and even artificial tissues.
- Specialized implants can be customized to meet patient needs, enhancing comfort and recovery outcomes.
- Custom surgical guides help doctors perform procedures with greater precision.
- Teams can quickly create test models before mass production.
- The production of small tools and components (such as pacemaker parts) is more convenient.
- Sensors can be directly printed on devices in certain fields to monitor health conditions.
3D printing technology supports rapid prototyping and customized production, suitable for both test models and final products.
CAD/CAM and 3D Imaging
CAD and CAM help turn your unique ideas into actual medical devices. NOBLE engineers use CAD to design devices tailored to patients. 3D imaging allows them to see all the details.
- CAD helps manufacture medical devices tailored to patients, such as dental implants.
- CAM machines carefully shape these designs.
- 3D modeling identifies issues before the first manufacturing run.
- This process ensures each device is properly fitted and functions effectively.
This technology makes work easier and more precise. It helps make the manufacture of medical devices safer and of higher quality.

CNC Machining
CNC machining plays an irreplaceable role in the manufacturing of medical devices. Computer-controlled tools are used to machine parts with extremely high precision. This machining method is widely used for various materials, such as metals and plastics.

CNC Milling
Because CNC milling can cut complex shapes and minute details, this process is particularly suitable for the machining of customized implants and tools.

EDM & Wire-Cuting
EDM and wire-cutting are capable of machining small, high-precision features. These methods are suitable for medical components requiring extremely high precision.
Swiss Machining
Swiss machining can produce small, high-precision components, such as parts for pacemakers. This process can efficiently repeat the same operations and produce components with smooth surfaces, making it one of our efficient and precise options.

CNC Routing
CNC routing shapes plastics and other materials. It is good for light device covers.
Manufacturing medical devices often requires significant capital investment, and CNC machining can greatly reduce error rates and material waste. Its precision is critical for safety and compliance. Companies like NOBLE often use it to customize medical devices and large projects for customers.

Injection Molding
Injection molding can quickly produce large quantities of medical devices. This method involves injecting molten plastic or rubber into a mold to form parts, enabling rapid production.

Plastic Injection Molding
This is used for mass production of sturdy plastic medical components such as syringes, caps, and test kits.

Insert Molding
Insert molding involves embedding metal or other materials into plastic components to enhance their strength.
Overmolding
Overmolding involves covering one material with another. When manufacturing medical devices, NOBLE uses this process to make handles, which helps improve the grip and comfort of handles or patches.
When we do injection molding, we use a bunch of safe materials to make sure each part is safe and high-quality. Companies like NOBLE use it for testing models and making a ton of medical parts.
Nanotechnology and Sensors
Nanotechnology works with tiny things to add new features. It makes devices stronger, flexible, and safer for the body. Nano-coatings stop germs from growing on implants. Nanoparticles help doctors find diseases sooner.
| Application Area | Description | Example Devices |
| Antibacterial Coatings | Tiny agents stop infections. | Nano-silver catheters |
| Diagnostic Nanoparticles | Quantum dots find disease signs. | Advanced test kits |
| Biosensors | Small sensors watch health all the time. | Glucose monitors, smart patches |
| Wound Healing | Nanofiber bandages help tissue heal faster. | Advanced wound care products |
| Regenerative Medicine | Nanomaterials help grow new tissues. | BioCartilage |
| Artificial Organs | Nanotech helps fake organs work better. | HeartWare™ Ventricular Assist System |
Sensors are now widely used in many new types of devices. They can monitor metrics such as heart rate and blood sugar levels. The Internet of Medical Things (IoMT) connects these sensors and transmits accurate data in real time to doctors and patients. Wearable devices, smart patches, and Bluetooth pacemakers can all use sensor technology to assist in medical care.
- 5G enables instant sharing of health data.
- Artificial intelligence analyzes sensor data to enable early warning.
- Cybersecurity measures ensure patient information security.
These new tools effectively help doctors detect abnormalities before problems worsen.
Note: As technology advances, robust cybersecurity measures are critical for protecting patient data.

Materials in Medical Device Manufacturing
When manufacturing medical devices, the process is an important element that NOBLE considers. At the same time, selecting the appropriate materials is also very important. This helps ensure patient safety and improve device performance. It also helps us comply with regulations from agencies such as the FDA. Each material has its own advantages and disadvantages. Engineers and designers must carefully weigh these factors before making a selection.
The following are the main steps our engineering team takes when selecting materials:
- Determine the device’s functions and usage.
- Identify the most critical performance metrics, such as strength or flexibility.
- Ensure the material is safe for human use and easy to clean.
- Test the material to verify its performance in actual use.
- Consider the material’s supply availability, production cycle, and cost.
- Regulatory compliance is always of utmost importance. Materials must meet stringent safety, biocompatibility, and sterilization requirements.
Medical-Grade Plastics
Medical-grade plastics are used a lot in healthcare. They are light, strong, and easy to shape. These plastics are used for syringes and wearable devices. Companies use them for prototypes because they are easy to test.
Some common plastics are:
- Polyethylene (PE)
- Polypropylene (PP)
- Polyvinyl chloride (PVC)
- Polycarbonate (PC)
- Acrylonitrile butadiene styrene (ABS)
- Polyetheretherketone (PEEK)
- Polymethyl methacrylate (PMMA)
- Polyamide (Nylon)
- Polymethylpentene (PMP)
Each type of plastic has its own specific uses. NOBLE’s engineers have compiled the following table to help you better understand how to select materials for medical device manufacturing:
| Medical Application | Common Medical-grade Plastics | Relevant Safety Properties |
| Medical Packaging | PE, PP, PVC | Non-permeability, sterilization resistance |
| Surgical Instruments | PP, PC, ABS | Biocompatibility, durability, and sterilization resistance |
| Implants | PE, PC, ABS, PEEK | Biocompatibility, lightweight, durability |
| Drug Delivery Systems | PVC, PP, PC | Non-permeability, pharmaceutical compatibility |
| Diagnostic Components | PS, PC, ABS | Transparency, durability |
| Dental Devices | PMMA, PC | Biocompatibility, transparency, and sterilization resistance |
| Wearable Medical Devices | PE, PC, ABS | Lightweight, durability, biocompatibility |
| Transparent Components | PMP | Transparency, low density, chemical resistance |
| High-temperature Applications | PEEK | High-temperature resistance, chemical resistance, and strength |
These plastics must be safe for human use and durable. They must also be able to withstand cleaning and disinfection. Companies use these materials in their factories for production tasks of varying sizes.
Metals and Alloys
Metals play an important role in manufacturing medical devices. When strength and durability are required, metals are widely used. Stainless steel, titanium alloys, and cobalt-chromium alloys are commonly used in implants and medical devices.
- Stainless steel is strong and does not rust. It is suitable for tools and implants.
- Titanium is lightweight and strong. It facilitates bone growth around it, making it suitable for joints and teeth.
- Cobalt-chromium alloys are tough and durable. Doctors use them for hip and knee joints.
- Nickel-titanium alloys can bend and return to their original shape, making them suitable for stents and guidewires.
Metals must be safe for the human body and not degrade inside it. Surface treatment technologies enhance compatibility between metals and tissues. CNC machining technologies can process these metals into complex components.
Tip: Selecting the appropriate metal materials can extend the lifespan of medical devices and enhance their performance.
Ceramics and Composites
Ceramics and composite materials have special applications in the manufacture of medical devices. Ceramics are characterized by their high hardness, stability, and safety for the human body. Doctors often use them to make bone replacements, dental crowns, and some heart valves.
Advantages of Ceramics:
- Extremely high strength and hardness
- Safe for the human body
- Can be implanted in the body for long periods of time or gradually dissolve over time
- Resistant to chemical corrosion and wear
Limitations of Ceramics:
- May crack when bent or impacted
- Not suitable for bending or stretching
- May fracture when small defects are present
Composite materials are a mixture of ceramics and plastics. They are strong and have good toughness. Some composite materials can promote the growth of new human tissue. These materials are often used in bone scaffolds and tissue engineering.
Engineers must select the appropriate ceramic or composite material based on the specific application. Some materials excel in strength, while others focus on promoting healing.
Biocompatible and Bioabsorbable Materials
Some medical devices remain in the body for extended periods of time, while others are designed to self-degrade after completing their intended function. Biocompatible materials do not harm the human body, while biodegradable materials safely degrade over time.
To ensure the safety of these materials, NOBLE conducts multiple tests:
- Assessing the degradation of materials within the body.
- Testing for the presence of potentially harmful chemicals that may be released.
- Adhering to strict regulations set by agencies such as the FDA and ISO.
- Conducting a comprehensive assessment of the entire device to ensure it is safe during manufacturing, cleaning, and use.
Common biodegradable materials include special plastics such as polylactic acid (PLA) and polyglycolic acid (PGA). These materials are used in sutures, bone screws, and drug delivery systems. Rigorous testing and standards help ensure the effectiveness of these materials and prevent harm.
Hydrogels, Textiles, and Batteries
New materials such as hydrogels, smart textiles, and batteries are transforming medical devices. Hydrogels are soft, flexible, and absorbent. They are ideal for wound care and sensors.
- Hydrogels can be stretched and bent. They are suitable for making skin-like sensors and patches.
- They can extend battery life and ensure safety. This is crucial for wearable devices.
- Some hydrogels can adhere to moist skin and continue to function even when sweating.
Textiles are also becoming smarter. Engineers are using them to create clothing that can monitor heart rate or movement. Such textiles must be comfortable, washable, and safe for the skin.
Batteries in some medical devices must be compact, lightweight, and reliable. New battery designs use hydrogels to enhance safety and flexibility. This helps power new wearable and implantable devices.
Simplicity and Safety in Manufacturing
Streamlined Processes
A medical manufacturing company can simplify production by adopting smart processes. Teams typically use lean principles to reduce waste and improve efficiency. NOBLE’s specific approach is as follows:
- Focus on the needs that matter most to customers.
- Review each step individually to identify and eliminate waste.
- Ensure that production processes run continuously to avoid stagnation.
- Produce only as needed, when needed.
- Continuously pursue improvement.
Many factories save time through automation, robotics, and rapid prototyping. For example, 3D printing technology helps teams quickly create prototypes to test designs before mass production. By partnering with advanced equipment and custom processing technology providers like NOBLE, companies can save years of time and significant costs. These measures help bring safe devices to market faster.
Regulatory Compliance
Regulations help ensure patient safety. In the United States, the U.S. Food and Drug Administration (FDA) sets rules for medical device manufacturing. Companies must comply with quality system regulations and obtain approval before selling new products. In Europe, companies must comply with the EU Medical Device Regulation and CE marking requirements.
Both regions inspect devices to ensure they meet strict standards and require regular inspections and documentation to prove safety. ISO 13485 is a globally recognized quality standard, and NOBLE adopts this standard to demonstrate compliance with quality requirements.
Quality Assurance
Quality assurance means that we inspect every stage of the manufacturing of medical devices to prevent errors. The NOBLE team uses systems like ISO 13485 to ensure each device meets high-quality standards. We train employees, use appropriate tools, and conduct regular inspections. This helps identify issues early and reduces the risk of defective devices reaching patients. Facilities that implement quality assurance have lower recall rates and better outcomes. They also use precision CNC machining and mold forming technologies to ensure the accuracy and safety of components.
Patient Safety
Patient safety is paramount. Design flaws, material errors, or improper use of medical devices can cause problems and even escalate into medical accidents. Sometimes, software failures or user errors can also cause problems. Equipment wear and tear or supply chain issues can also pose risks. To ensure patient safety, companies like NOBLE test their equipment, continuously monitor issues after market release, and address them promptly. They implement strict quality control measures and learn from each case to ensure that every piece of equipment can serve patients safely and effectively.
Best Practices for Medical Device Manufacturing

Design for Manufacturability
Design for Manufacturability (DFM) helps teams design medical devices that are easy to manufacture, safe, and cost-effective. When engineers and designers work closely together in the early stages, they can quickly identify issues and avoid major changes later on. Here are some best practices:
- Use concurrent engineering to enable teams to plan manufacturing processes while designing products. This helps save time and costs.
- Ensure that design transfer steps comply with FDA and ISO 13485 standards.
- Develop components and assembly steps in parallel during the design phase to identify risks early on.
- Focus on using reliable materials, workers, and equipment.
- Experiment with rapid prototyping and additive manufacturing technologies to quickly validate design concepts.
- Consider future manufacturing options in advance to avoid project delays.
- Collaborate with manufacturing engineers and suppliers who understand clinical requirements.
- Use simulation tools to validate designs before actual manufacturing.
- Comprehensively document design and manufacturing data.
- Carefully select materials and verify their safety and compliance.
- Encourage collaboration and respect among team members.
If a team neglects DFM, costs may rise, projects may be delayed, and regulations may be violated. Companies that follow these steps can produce safer products and deliver them to users more quickly.

Quality Control
Quality control in medical device manufacturing differs significantly from other industries. Teams must strictly adhere to relevant regulations such as the FDA QSR (Quality System Regulation) and ISO 13485. We utilize advanced equipment, such as precision CNC machining and 3D printing technology, to ensure that every medical component meets quality standards. Quality control encompasses every stage of the production process, from selecting safe materials to testing finished devices.

The factory provides comprehensive training for employees and employs new methods to test the safety and functionality of devices. Even after devices are sold, the team continues to monitor their usage to identify any potential issues. This emphasis on safety and compliance not only safeguards patient safety but also enhances public trust in the devices.
Supplier Traceability
Supplier traceability means knowing the origin of each component and its final destination. Medical device companies keep detailed records at every stage. They typically work with electronic contract manufacturers who use verified suppliers and are ISO 13485 certified. Engineers conduct on-site inspections of components and collaborate with suppliers to ensure compliance.
Many companies use barcodes, QR codes, and RFID tags to track components in real time. Real-time location systems (RTLS) help monitor devices during storage and transportation. These tools assist companies in adhering to regulations and facilitating recalls of non-compliant medical devices when necessary.

Your Trusted Medical Equipment Manufacturer – NOBLE
NOBLE is a leading medical manufacturing company in China. Its engineers are highly skilled, and its service staff are friendly. They assist customers in turning their ideas into actual products. NOBLE can produce single prototypes or large-scale products. They use advanced equipment and customized processes, enabling them to deliver results quickly and safely.
Advanced Technology
NOBLE employs the most advanced technology in the field. Its facilities are equipped with precision CNC machining and mold manufacturing equipment. For complex equipment, they utilize specialized processes. The table below highlights the key technologies and processes that set NOBLE apart:
| Technology/Process | Description |
| X-STATIC® and CIRCUITEX® | Uses pure silver in yarns and fabrics for FDA-cleared devices. |
| Regulatory Compliance | Has many FDA 510(k) clearances and meets EU rules. |
| Quality Management | ISO 9001:2015 certified and uses Six Sigma to improve. |
| Statistical Process Control (SPC) | Watches and checks production to keep quality high. |
| Analytical Testing | Uses labs for chemical and microbiology testing. |
| Physical Property Testing | Checks strength, flexibility, and conductivity with ASTM standards. |
| Material Recovery and Recycling | Recovers silver and treats waste to help the environment. |
NOBLE assists with risk assessment, user research, and rule-making planning. Its engineers work with customers to jointly design and test new products. We also provide training, operating guidelines, and product launch support. This makes the entire process more convenient for all parties involved.
Quality Commitment
NOBLE always puts product quality first. We have obtained ISO 13485 certification. Our process validation department has conducted more than 1,700 inspections on Class II and Class III implants and medical devices. The team strictly controls every process step from IQ/OQ to risk analysis to ensure that every product meets stringent quality standards.
NOBLE continuously optimizes its quality management system. The company has passed rigorous audits by the British Standards Institution (BSI) and has maintained its certification status since 2004. This fully demonstrates its commitment to complying with the evolving regulatory requirements of the U.S. Food and Drug Administration (FDA), Europe, and other regions. Its production facilities utilize CNC machining, laser cutting, and welding technologies to ensure precise component fit. Rapid prototyping and scalable production capabilities enable customers to obtain products faster. NOBLE can produce single prototypes or large-scale production batches.
NOBLE’s relentless pursuit of quality, technology, and service makes it a trusted partner for customers in the medical device manufacturing industry, providing them with safe and convenient solutions.
FAQ for Medical Manufacturing
What does a medical manufacturing company do?
A medical manufacturing company makes devices for healthcare. They design and build products for doctors and patients. Workers use special machines like CNC machining and molding. Every product must follow safety rules. The company checks that each device works well.
Why is a prototype important in medical device manufacturing?
A prototype helps engineers test their ideas first. Teams can find problems before making lots of devices. Factories use prototypes to see if designs work with real equipment. This step helps make sure custom processes are correct.
How does molding help in making medical devices?
Molding shapes plastic or rubber into device parts. It is quick and makes many pieces that look the same. Companies use molding to make strong, safe parts for tools and covers.
What is precision CNC machining used for?
Precision CNC machining shapes metal or plastic very carefully. Factories use it for small, detailed parts in implants and tools. This process keeps every part the right size and shape.
How do companies keep medical devices safe?
Companies check quality at every step. They test materials and watch how things are made. Teams use advanced equipment and follow safety rules. Problems are fixed quickly to keep patients safe.








